
KIIT-TBI Ecosystem with the support of
BIOTECHNOLOGY INDUSTRY RESEARCH ASSISTANCE COUNCIL (BIRAC)
About: BIOTECHNOLOGY IGNITION GRANT
Biotechnology Ignition Grant (BIG), a flagship scheme of BIRAC, DBT, GoI. is India's largest early-stage biotech funding programme supporting ideas from start-ups and individual entrepreneurs. The ideas should have clear potential to translate into commercial products/technologies for which the scheme provides grant-in-aid of up to INR 50 Lakhs (approx USD 75,000).
Objectives: BIOTECHNOLOGY IGNITION GRANT
◉ Foster the generation of ideas with commercialization potential from and for North East.
◉ Upscale and validate proof of concept
◉ Encourage researchers to take technology closer to market through a start up
◉ Stimulate Entrepreneurship to enable translation of ideas into commercially viable products/ technologies which address an unmet need
Duration: BIOTECHNOLOGY IGNITION GRANT
18 months
Eligibility: BIOTECHNOLOGY IGNITION GRANT
◉ Individuals (Students, Academicians, Scientists, Clinicians, Researchers)
◉ Biotech start ups (less than 5 years old)
Funding: BIOTECHNOLOGY IGNITION GRANT
Grant-In-Aid Upto INR 50 Lakhs
Impacts: BIOTECHNOLOGY IGNITION GRANT
Technology Supported
IPs CREATED
Product Commercialised
Follow on Funding
Funds Mobilised
Women led Startups
TImeline: BIOTECHNOLOGY IGNITION GRANT
Preliminary Selection Comittee
Scrutiny by BIG Partner for the eligibility Criteria under BIG Scheme
Adequate technical Details to be Scored by Different subject experts
Expert Panel
One-to-one presentation before the Technical Expert Panel
Committee
Final Decision with respect to a Cut off score across different themes
Thematic Area: BIOTECHNOLOGY IGNITION GRANT

Agriculture

Biosimilars &
Stem Cells

Drugs Including
Drug Delivery

Industrial
Biotechnology

Devices &
Diagnostics
Vaccines

Bioinformatics &
Facilities

Other Emerging Technology
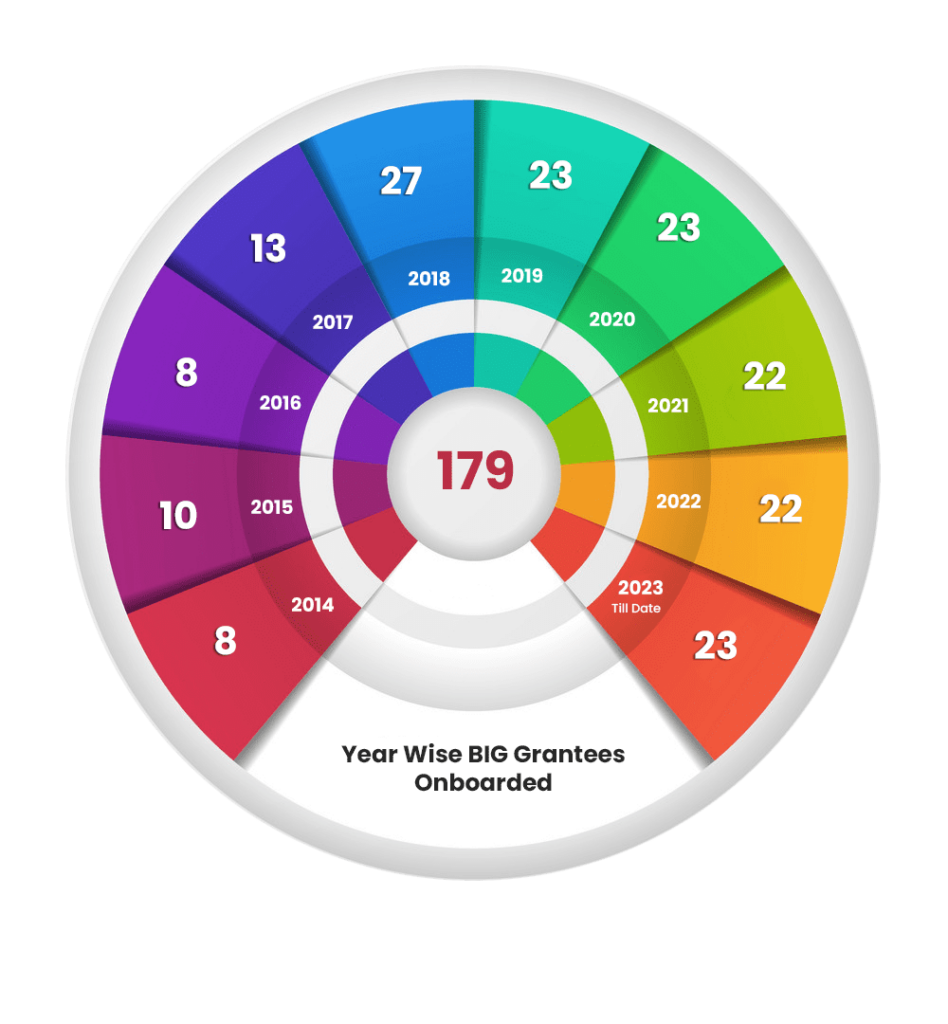
The Big Journey of kiit-tbi
Total Applications Received: 2700
Startups 46.8%
individuals 53.2%
Total Grants Sanctioned : 179
Startups 75.9%
individuals 24.1%
- Healthcare Devices:20%
- Agriculture: 13.9%
- Diagnostics: 20.6%
- Industrial Biotechnology: 29%
- Drugs & Vaccines: 16.5%
BIOTECHNOLOGY IGNITION GRANT: Benefits
Grant-In-Aid support up to INR 50 lakh For the ideation to PoC.
Tailored mentoring in areas like IP, Legal, Regulatory, Business Development.
Opportunity to pitch for follow-on funding under other KIIT-TBI's programs.
Access the digital fabrication lab to turn your ideas into prototypes.
Know more...
Domain Specific Personalized mentoring & Networking Support
Access the digital fabrication lab to turn your ideas into prototypes.
BIOTECHNOLOGY IGNITION GRANT: Grantees
- All
- Call-4
- Call-5
- Call-6
- Call-7
- Call-8
- Call-9
- Call-10
- Call-11
- Call-12
- Call-13
- Call-14
- Call-15
- Call-16
- Call-17
- Call-18
- Call-19
- Call-20
- Call-21
- Call-22
- Call-23
BIOTECHNOLOGY IGNITION GRANT: Mentors




































faq
Biotechnology Ignition Grant (BIG) Scheme is grant-in-aid funding to support individuals and startups in the field of Biotechnology. BIG scheme invests in ideas that have clear potential to translate into commercial products/technologies. The scheme provides funding, mentorship, access to incubation, other supports for ideation to proof of concept. Funding will be in the form of Grant-in-Aid up to INR 50 Lakhs. So, if you have an idea, strong rationale supporting it, addressing an unmet need, you can apply for BIG funding to progress from ideation to Proof-of-concept.
Healthcare, Life sciences including Drugs, Biologics, Delivery systems, Medical Devices, Diagnostics, Biomaterials, others; Industrial Biotechnology including enzymes, biofertilizers, bioprocesses, waste treatment, others; Agriculture and allied areas such as Nutrition, Fisheries, Poultry, Sericulture, Animal Husbandry, Aquaponics, others; Waste to value; Sanitation; Biofuels, Clean Energy; Bioservices and Bioanalytics including Big Data, Artificial intelligence, Machine Learning, Internet of Things, Automation integrated in Biotech applications.
BIG award provides funding support for buying equipment, reagents, components, hiring skilled manpower for team building, travel, incubation charges, rentals, engaging services, etc in the 18 months of BIG grant.
During the BIG tenure, BIRAC extends several networking opportunity to connect with experts, mentors, industry, investors, peer-to-peer learning, specialized trainings & workshops, exposure to various platforms on merit basis. These opportunities are made available through BIRAC’s ecosystem supporting partners including BIG partners, BioNEST clusters, and a huge network of extended partners.
Yes, if you are pursuing translational research leading to formation of a startup. As a procedural requirement, you are required to obtain a No Objection Certificate (NOC) from your institute/employer. Faculty members who are not willing to create a Startup within BIG term of 18 months are encouraged to apply under other schemes of BIRAC for Faculty.
Yes, you can apply as an individual but need to provide an undertaking that you will terminate your association with the current employer and take up the project full time if selected.
Or, your application should be submitted under Company/LLP category with the employer company being the applicant company.
The indicative criteria for evaluation of an application include Unmet need, Value proposition, Technical viability, Team strength. Proposed product/ technology must have Commercialization Potential.
It is 4 tier selection process. All eligible applications go through the following steps:
- Preliminary Selection Committee (PSC) screens the proposals for scheme fit, adequacy of details, plagiarism
- The PSC recommended proposals are then reviewed Online by subject experts
- The recommended applicants are then called for F2F presentations before Technical Evaluation Panel (TEP). The proposal will be scored on a scale of 100.
- Expert Selection Committee (ESC) will decide the final cutoff of marks and recommend the proposals for BIG funding.
It typically takes 4 months for the final announcement of BIG results from closing of the call. You will be updated at each stage by BIG Partners about your application status.
No, but only ONE will be eventually funded. It is encouraged to submit the best one proposal (the most prioritized) by the applicant. Submitting more than one project reflects poorly on the applicant’s ability to prioritize options.
Incubator is a center that has the required lab infrastructure, required network to support your proposed BIG work. Certain BioNEST incubators have been officially recognized as BIRAC’s BIG Partners. These incubators are mandated to implentthe BIG Scheme (Facilitate and mentor the aspirant applicants and BIG grantees). BIG Partners are responsible for initial screening of the proposal, technical due diligence, financial due diligence, project monitoring, fund disbursement, arranging for workshops etc.
The applicant will receive the fund in a separate dedicated, auditable no-lien account. The fund disbursement is linked with milestones through BIRAC’s BIG Partners.
BIG grantees on successful progression to commercialization are required to pay 5% royalty on net sales of the product/technology developed through BIRAC support till the time such royalty payment becomes equivalent to the grant-in aid assistance disbursed by BIRAC for the Project.
Confidentiality is maintained for all the proposals that are received under different schemes. All the external reviewers sign a non-disclosure agreement and no conflict agreement before viewing the proposals.




